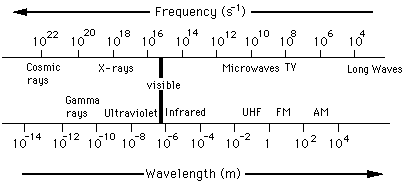
 |
 |
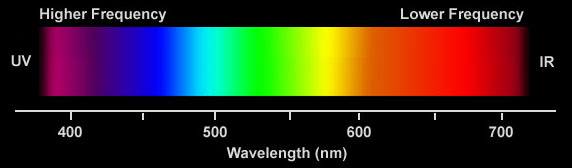
|
Red + Yellow makes Orange Yellow + Blue makes Green Blue + Red makes Violet |
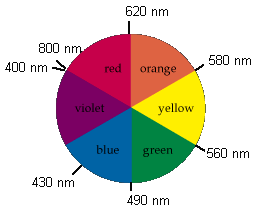
Example:
|
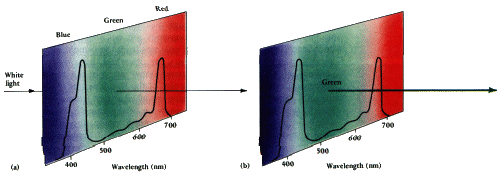
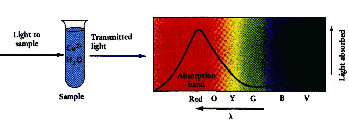
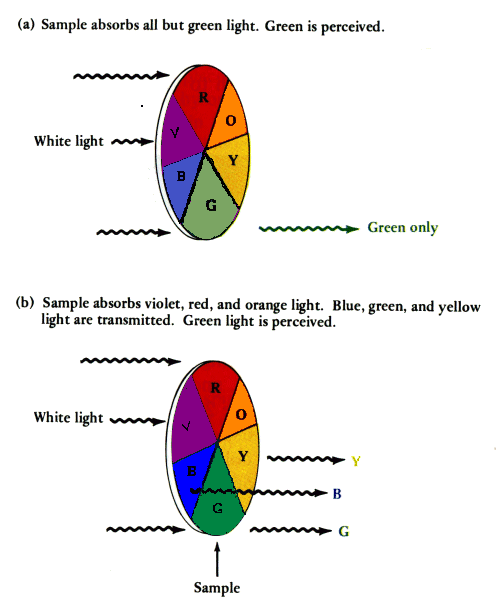
If red, yellow,
orange, blue and
violet are absorbed... only one color is transmitted ... |
If violet, red,
and orange are absorbed... blue, green , and yellow are transmitted ... the middle color is perceived... |
 |