The negative value of the Enthalpy of Reaction indicates that the reaction is a heat producing or exothermic.
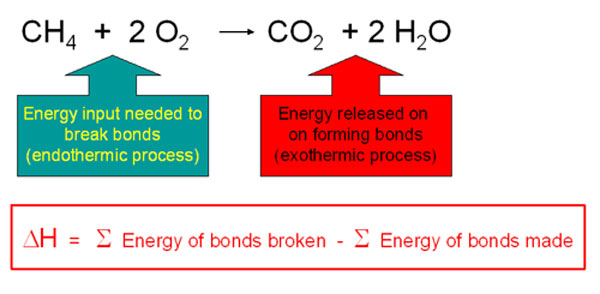
During chemical reactions, energy is either released to the environment (exothermic reaction) or absorbed from the environment (endothermic reaction). During chemical reactions, bonds are broken in the reactants and new ones are made in the products. Bond-breaking is an endothermic process and bond-making is an exothermic process. The average bond dissociation energies of some chemical bonds are shown in the following table.
|
Selected
Bond Energies
|
|||
| Bond | Bond Energy(kJ/mole) | Bond | Bond Energy(kJ/mole) |
| H-H | 432 | C=O | 799 |
| O=O | 494 | C-C | 347 |
| O-H | 460 | C=C | 611 |
| C-H | 410 | C=C (aromatic) | 519 |
| C-O | 360 | N=O | 623 |
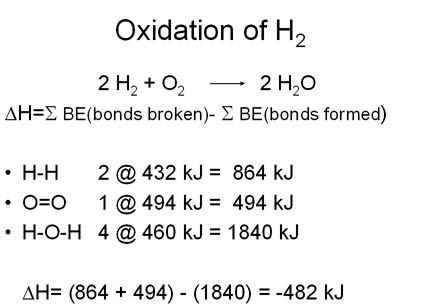
For any chemical reaction, the overall energy change, the enthalpy of reaction(DH), is the difference of all the energy absorbed in bond-breaking and all the energy released in bond-making.
DH = S BE(bonds broken)- SBE(bonds formed)
|
The negative value of the Enthalpy of Reaction indicates that the reaction is a heat producing or exothermic. |
The energy produced in exothermic reactions can be used directly or be converted to other energy forms.
An oxidation-reduction or redox reaction is one in which electrons are transferred between two species. The rusting of iron is an example of a redox reaction.
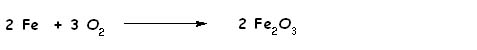
In this reaction iron metal losses electrons (is oxidized) and the oxygen gains electrons (is reduced).
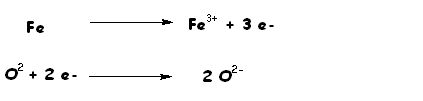
In a redox reduction something is always oxidized while another species is reduced.
Oxidation is easy to recognize when an element changes changes oxidation state to become an ion. However when dealing with organic molecules, atoms are joined by covalent bonds rather than ionic ones so oxidation is not as easily recognized.
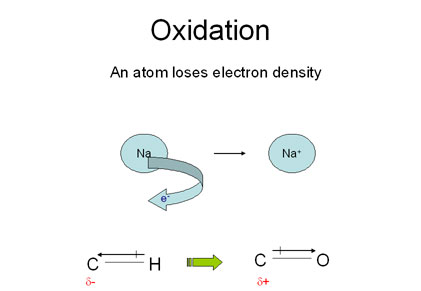
In organic molecules, an atom is oxidized if during a reaction it becomes bonded to a more electronegative element which pulls electron density away from it.
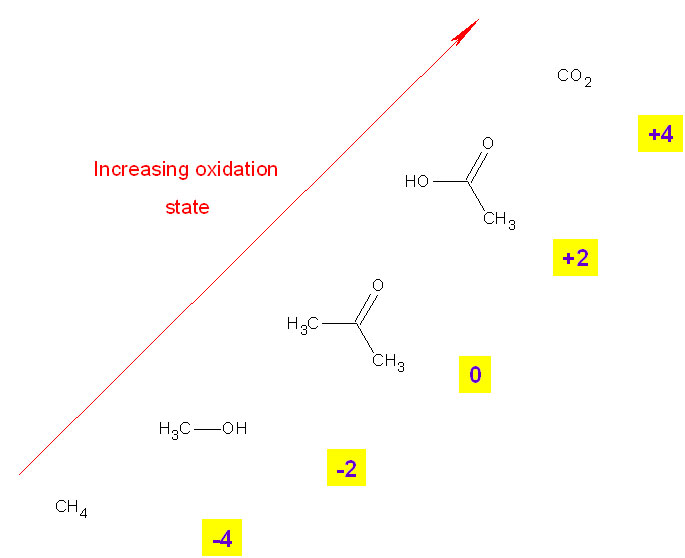
In general, oxidation occurs if the oxygen content of a covalently bonded molecule increases or if the hydrogen content decreases.The carbon atom in methane (CH4) is more reduced than the carbon atom in carbon dioxide (CO2)
Combustion is a reaction with oxygen. In the case of the combustion of fossil fuels, the combustion reaction is what we think of as a burning process. In the combustion reaction, the species reacting with the oxgyen is oxidized (because oxygen is very electronegative). Fossil fuels are composed primarily of hydrocarbons (molecules containing primarily carbon hydrogen bonds). In these molecules carbon is in a very reduced state. During the combustion reaction, the hydrocarbon molecules are converted to carbon dioxide and water.
![]()

Every mole of methane (16 g) releases 810 KJ of energy on burning.
Combustion energetics can be estimated from the bond energies for all the classifications of fossil fuels. The amount of energy released is dependent on the oxidation state of the carbons in the hydrocarbon which is related to the hydrogen/carbon ratio. The more hydrogen per carbon, the lower the oxidation state and the more energy that will be released during the oxidation reaction. Thus the greater the H/C ratio, the more energy release on combustion.
Petroleum has a large saturated hydrocarbons content. For estimating energy content, we can consider the molecule to consist of multiple -CH2-units. For calculating the expected energy release per CH2 unit, the energy of two C-H bonds and one C-C bond (since the carbon joins two neighboring groups and we are averaging) would be used which gives an energy content similar to that of methane ("natural gas"). However, petroleum also can contain a significant fraction of aromatic molecules depending on the nature of the petroleum distillate product. For example crude oil has an energy content of about 45.2 kJ/g while gasoline has a higher value of about 48.1 kJ/mole because it contains a smaller fraction of aromatics.
Coal is composed of primarily aromatic hydrocarbons, so we can consider the molecule to consist of multiple -CH- units. The actual average energy release per gram of coal from combustion is less than the predicted value since coal contains significant amounts of water and minerals. Hard coals such as bituminous or anthracite have larger energy content (29-33 kJ/g) than the soft sub-bituminous or lignite coals (17-21 kJ/g).
![]()

The average hydrogen/carbon ratios show that the degree of unsaturation increases from natural gas through petroleum to coal. The amount of carbon dioxide released per mole also increases as the amount of unsaturation increases. Since carbon dioxide is a greenhouse gas, from this data, it would appear that burning coal would have a larger greenhouse effect than burning natural gas.

Ethanol (CH3CH2OH, a biomass derived fuel) is more highly oxidized than a hydrocarbon since it contains an oxygen and gives off significantly less energy on combustion than petroleum. Gasohol is a gasoline/ethanol mixture.
|
Ethanol vs
Petroleum
|
|
|
|
|
1. The major component of natural gas is methane while the gas used in your barbecue is propane (C3H8). Assuming complete, clean burning, compare the amount of energy released by each in terms of kJ/mole of carbon dioxide produced and in terms of kJ/g of fuel. Do your calculations based on based on bond breakind and making that occurs in the combustion reaction. Hint count up the number of C-C and C-H bonds in the balanced equation for each combustion reaction. Propane has the formula CH3CH2CH3. 2. Compare the costs per unit of energy for
electricity and gasoline. Assume $0.06 per kWh for electricity and $1.50
per gallon for gasoline. Make the same comparison for gasoline at $2.35/gal.
Since gas engines are only about 20-25% efficient and elecric cars are
50-80% efficient, which is more economical for transport with respect
to fuel costs? If the electricity to charge the electric car's battery
is obtained from a coal-fired plant which operates at an efficiency of
33%, how does this change the picture? 1 lb gasoline releases= 19,000 BTU/lb 1 gal gasoline= 5.51 lb 1 cal=4.18 j = 1.16 x 10-6KWh =3.97 x 10-3BTU 3. Petroleum consumption in the United States in 1999 was 39.8 EJ with about 60% (23.8 EJ) coming from foreign sources. Transportation consumed 66% (26.2 EJ) of the total. Passenger cars (~132 million) consumed 9.6 EJ while light trucks (~72 million vans, pickups and SUVs) consumed 6.7 EJ. If the average fuel efficiency of a passenger car was 21.4 mpg and a light truck 17.1 mpg, determine how much less petroleum would have been imported if these vehicles had been replaced by hybrid cars (fuel efficiency 60 mpg) and hybrid light trucks (fuel efficiency 40 mpg). |