Oxides
Basic Oxides
The oxide ion is a very strongly basic anion due to its very small size and high charge. Since hydroxide ion is the strongest base that can persist in water, the oxide ion reacts quantitatively with water to generate hydroxide ions. This is an example the leveling property of very strong bases.As predicted by the generalized solubility rules, only those metal oxides of the nonacidic and feebly acidic cation dissolve in water. The dissolution is highly exothermic generating the hydroxide of the cation. These aqueous solutions are strongly basic. Metal oxides which exhibit this behavior are termed basic oxides because they act as bases. The oxides of nonacidic cations are so water reactive that they are seldom seen. These oxides cannot be prepared by dehydrating the hydroxide at high temperature.
The oxides of feebly acidic cations are more common. Lime, CaO is an example. Lime can be prepared comercially by the thermal decomposition of limestone.
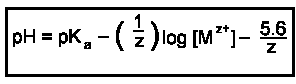
The oxides of feebly acidic cations react exothermically with water producing the hydroxide.
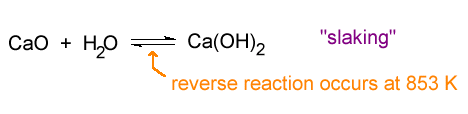
The hydroxides of feebly acidic cations are not deliquescent.
The oxides of weakly acidic cations and moderately acidic cations are insoluble in water. These oxides do not introduce significant amounts of O2- ion into the solution so hydroxide ions are not produced. Although these oxides do not significantly alter the pH of the water, they are still bases and neutralize strong acids.
Acidic Oxides (Acid Anhydrides)
Many of the covalent oxides of the very acidic cations (hypothetical) exhibit acidic properties. Many of these oxides dissolve in water to give the oxo anion in which the element has the same oxidation number as the oxide. The oxide will be soluble if its reaction with water produces a strong or very strong acid because these acids ionize completely shifting the equilibrium toward dissolution. If the reaction with water produces a moderately acidic oxoacid, the oxide may or may not be soluble. If the oxoacid generated is weakly acidic, the oxide is usually but not always insoluble in water.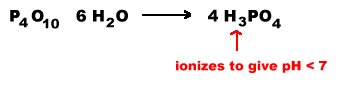
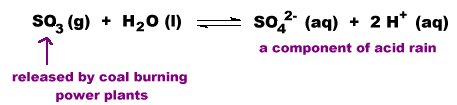
Some acidic oxides such as those of sulfur and nitrogen are air pollutants because they react with moisture in the air to produce acid rain.
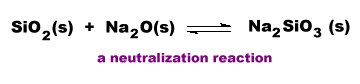
There are a few oxides, such as NO2 and ClO2 in which the central atom's oxidation state does not correspond to the oxidation state of that element in a stable or known oxo acid. Such oxides generate a mixture of oxo acids or anions by disproportionation.
Amphoteric Oxides
Some oxides exhibit both acidic and basic properties.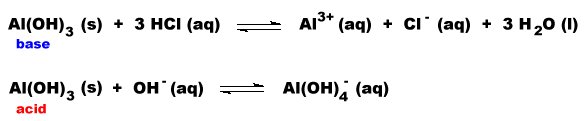
General Rules
In general, the electropositive character of the oxide's central atom will determne whether the oxide will be acidic or basic. The more electropositive the central atom the more basic the oxide. The more electronegative the central atom, the more acidic the oxide. Electropositive character increases from right to left across the periodic table and increases down the column.
| Li | Be | B | C | N | O | F |
| Na | Mg | Al | Si | P | S | Cl |
| K | Ca | Ga | Ge | As | Se | Br |
| Rb | Sr | In | Sn | Sb | Te | I |
| Cs | Ba | Tl | Pb | Bi | Po | At |
There are three nonmetal oxides from the upper right portion of the periodic table, CO, NO, and N2O, which have such low oxidation numbers for the central atom that they give neutral aqueous solutions.
Since the acidity of a cation rises rapidly with charge, d-block elements which exhibit a wide variety of oxidation numbers may have one or more oxides that exhibit only basic properties and one or more oxides that exhibit only acidic properties. The higher the oxidation number the more acidic the corresponding oxide. Chromium is an example of such an element.
| Oxide | Oxidation Number | Category |
| CrO | Cr2+ | basic |
| Cr2O3 | Cr3+ | amphoteric |
| CrO3 | Cr6+ | acidic |
Basic oxides react with acidic oxides to produce salts of oxo anions.
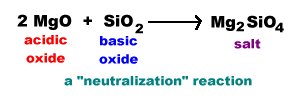
Since there is no water involved salts of oxo anions which are too basic to persist in water can be formed. These reactions of acidic and basic anions have important practical applications such as in the control of gaseous acidic oxides that when released into the atmosphere result in acid rain.
These reactions are also used in the production of materials such as concrete, glass and ceramics.
 Complete and balance the Complete and balance the following chemical equations for reactions of oxides. |